The presence of E. coli bacteria indicates hygiene issues
Microorganisms in water systems, particularly pathogenic bacteria, threaten public health [1]. Waste from humans and animals might cause contamination of water, where Escherichia coli (E. coli) bacteria indicate fecal contamination since they are found almost exclusively in human and animal feces [1].
An indicator for fecal pollution is needed for public health protection [2] and this is precisely where E. coli comes into play [3]. E. coli hence takes on an important role in hygiene indication [4] and is considered the best biological drinking water indicator to ensure public health [2]. The presence of E. coli strains, especially pathogenic ones, points out water quality and hygiene issues [4] [5].
Using E. coli contamination as an indicator is therefore a cost-effective way of examination of food and water for protection of public health [2] without testing for a whole range of pathogens in separate tests. E. coli possibly indicates presence of other foodborne pathogens [6] such as Salmonella, Listeria, Shigella, Vibrio, Hepatitis A [7].
Pathogenic E. coli O157 remain undetected by gold standard method
E. coli are gram-negative, rod-shaped bacteria residing in the gastrointestinal tract of humans and animals [8]. While most strains are harmless, there are some pathogenic strains which are distinguished e.g. based on clinical symptoms or serogroups [8].
Lipopolysaccharides on the surface of E. coli cells make up O antigens which determine the O serogroup and are crucial for host-pathogen interaction [9]. For instance, E. coli O157:H7 possesses the somatic O antigen 157 and the flagella (H) antigen 7 [8]. This specific strain (E. coli O157:H7) is one of the most serious foodborne pathogens causing severe disease and thereby posing a significant threat to public health [8]. In the US for instance, E. coli O157:H7 is most frequently transmitted through consumption of contaminated food and water [8]. Due to the lack of a specific treatment for E. coli O157:H7, prevention and control are even more critical [8].
An infection with E. coli O157 can become very dangerous since it might cause haemolytic uraemic syndrome which is the most frequent cause of acute renal failure in children in the Americas and Europe [10]. Unfortunately, there are no treatment options available to reduce the risk of developing the haemolytic uraemic syndrome [10].
Almost all E. coli clinically isolated generate beta-D-glucoronidase which renders this enzyme useful for detection of E. coli in water, food as well as environmental samples where these bacteria indicate possible fecal contamination [11]. However, we need to be careful using beta-D-glucoronidase for fecal E. coli detection [11] since E. coli O157:H7 is not capable of producing beta-glucoronidase [8]. Because E. coli O157 is beta-glucoronidase negative, it will not be detected as E. coli by the ISO plating method (ISO 9308-1:2014).
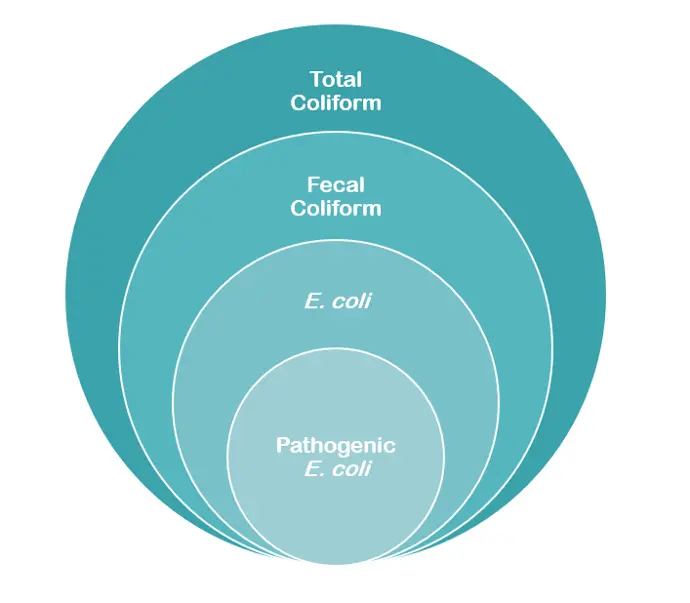
Chart: From Total Coliform bacteria to pathogenic E. coli.
Pathogenic, Shiga toxin-producing E. coli (STEC) are most frequently involved in foodborne outbreaks
and the CDC estimates
that STEC O157 is responsible for approximately 36% of STEC infections in the
United States. The gold standard ISO-method performed on petri dishes however fails to detect E. coli O157. Detection with the rqmicro method does not depend on enzymes and therefore also detects the particularly dangerous serotype E. coli O157.
Benefit from market-leading time to result and detection of pathogenic strains with rqmicro's E. coli Test Kits
Investigating the microbiological drinking water quality requires testing for indicator bacteria such as E. coli which is the best bacterial indicator for fecal contamination in drinking water [5]. The detection and quantification of E. coli in water is crucial to identify fecal contaminations and to keep water safe [1].
E. coli O157:H7 is responsible for many disease outbreaks which is why it is so important to have a rapid and reliable detection method at hand [12]. Early recognition of contamination is crucial for the food industry [13] and might prevent product recalls [1]. The method of detection needs to be highly sensitive since E. coli bacteria might only be present at very low levels [1]. The cultivation-based detection method according to ISO 9308-1:2014 fails to detect the particularly dangerous serotype O157 of E. coli and requires 24 to 72 hours to render results [1]. Although faster techniques (PCR, ELISA) are available, they are expensive, require sample transport to the laboratory and involve complex equipment and procedures which can only be handled by trained professionals [1].
The easy-to-use rqmicro E.coli test is based on flow-cytometry technology, includes the particularly dangerous serotype E. coli O157 and delivers results during the same shift. Depending on requirements, users opt for a qualitative result with lowest detection limit (LOD) of 1 cell / 100 mL available after 8 hours, or a quantitative result with a limit of detection (LOD) of 10 cells / 100 mL in a market-leading time to result of 2 hours.
Learn more about our two different E. coli test kits on our website.
The portability of the rqmicro.COUNT instrument allows
you to directly visit critical control points in the food and beverage industry
with it. As a result, rqmicro.COUNT will
facilitate your risk management and enable you to make water and food products
safe for consumption.
Get in touch with rqmicro experts for a free consultation on our solution.
References:
[1] Nurliyana, M. R., et al. "The detection method of Escherichia coli in water resources: A review." Journal of Physics: Conference Series. Vol. 995. IOP Publishing, 2018.
[2] Edberg, S. C. L., et al. "Escherichia coli: the best biological drinking water indicator for public health protection." Journal of applied microbiology 88.S1 (2000): 106S-116S.
[3] Keshav, V., et al. "E. coli from dishcloths as an indicator of hygienic status in households." Journal of Water, Sanitation and Hygiene for Development 5.3 (2015): 351-358.
[4] Scheinberg, Joshua A., et al. "Prevalence and phylogenetic characterization of Escherichia coli and hygiene indicator bacteria isolated from leafy green produce, beef, and pork obtained from farmers' markets in Pennsylvania." Journal of food protection 80.2 (2017): 237-244.
[5] Odonkor, Stephen T., and Joseph K. Ampofo. "Escherichia coli as an indicator of bacteriological quality of water: an overview." Microbiology research 4.1 (2013): e2.
[6] Truchado, Pilar, et al. "Correlation between E. coli levels and the presence of foodborne pathogens in surface irrigation water: establishment of a sampling program." Water research 128 (2018): 226-233.
[7] Bintsis, Thomas. "Foodborne pathogens." AIMS microbiology 3.3 (2017): 529.
[8] Lim, Ji Youn, Jang W. Yoon, and Carolyn J. Hovde. "A brief overview of Escherichia coli O157: H7 and its plasmid O157." Journal of microbiology and biotechnology 20.1 (2010): 5.
[9] DebRoy, Chitrita, Elisabeth Roberts, and Pina M. Fratamico. "Detection of O antigens in Escherichia coli." Animal health research reviews 12.2 (2011): 169-185.
[10] Pennington, Hugh. "Escherichia coli O157." The Lancet 376.9750 (2010): 1428-1435.
[11] Chang, GEORGE W., J. A. N. I. N. E. Brill, and R. O. S. A. L. I. N. D. Lum. "Proportion of beta-D-glucuronidase-negative Escherichia coli in human fecal samples." Applied and Environmental Microbiology 55.2 (1989): 335-339.
[12] Deisingh, A. K., and M. Thompson. "Strategies for the detection of Escherichia coli O157: H7 in foods." Journal of applied microbiology 96.3 (2004): 419-429.
[13] De Boer, E., and A. E. Heuvelink. "Methods for the detection and isolation of Shiga toxin‐producing Escherichia coli." Journal of applied microbiology 88.S1 (2000): 133S-143S.